
Topic 1: The changing models of atomic structure 
Describe the contribution of protons and neutrons to atomic nuclei in terms of the proton number and nucleon number (ii) distinguish between isotopes on the basis of different numbers of neutrons present (iii) recognise and use the symbolism y xA for isotopes, where x is the nucleon number and y is the proton number.Deduce the numbers of protons, neutrons and electrons present in both atoms and ions given proton and nucleon numbers.Describe the distribution of mass and charges within an atom.Identify and describe protons, neutrons and electrons in terms of their relative charges and relative masses. 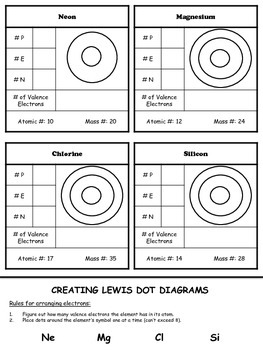
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
